
Goal
The goal of this site is to provide the best information we can, focused around safety, on automating a manual resuscitator, as a potential means for longer-term ventilation. This is a completely off-label use, but we recognize the global interest when a hospital has used up all ventilators and the only option is manual bagging a patient. We hope that such systems may serve as bridge devices and help with the triage of available respirators and clinicians trained in respiratory therapy. This may allow less severe patients to be cared for by less specialized clinicians, while resources are focused on those most in need. However, at no time should a patient be unattended without someone skilled available to directly monitor their vital signs. Effectively, we are reprising the early days of safe ventilation where direct clinical observation of patient condition served as the key feedback.
Begin by reading the Key Ventilation Specifications, then the detailed clinical information. This is critical to understand the logic underlying the mechanical, electrical, controls and testing information. We have just posted essential safety information on removing dead space.
Background & Need
We are one of several teams who recognized the challenges faced by Italian physicians, and are working to find a solution to the anticipated global lack of ventilators. In the US alone, the COVID-19 pandemic may cause ventilator shortages on the order of 300,000-700,000 units (CDC Pandemic Response Plans). These could be present on a national scale within weeks, and are already being felt in certain areas. An increase in conventional ventilator production is very likely to fall short and with significant associated cost (paywall warning).
Almost every bed in a hospital has a manual resuscitator nearby, available in the event of a rapid response or code where healthcare workers maintain oxygenation by squeezing the bag. Automating this appears to be the simplest strategy that satisfies the need for low-cost mechanical ventilation, with the ability to be rapidly manufactured in large quantities. However, doing this safely is not trivial.
Use of a bag-valve mask (BVM) in emergency situations is not a new concept. A portable ventilator utilizing a manual resuscitator was introduced in 2010 by a student team in the MIT class 2.75 Medical Device Design (original paper here and news story here), but did not move past the prototype stage. Around the same time, a team from Stanford developed a lower-cost ventilator for emergency stockpiles and the developing world. It looks similar to a modern ICU ventilator (Onebreath), but “production for US hospitals would start [in] about 11 months”, making it “a second wave solution” (MIT Tech Review Article). Last year, the concept was re-visited by two student teams, one from Rice university (here & here), and another Boston-based team who won MIT Sloan’s Healthcare prize (MIT News: Umbilizer). Other teams currently working on this challenge can be found linked on our Resources page.
Key Research Question
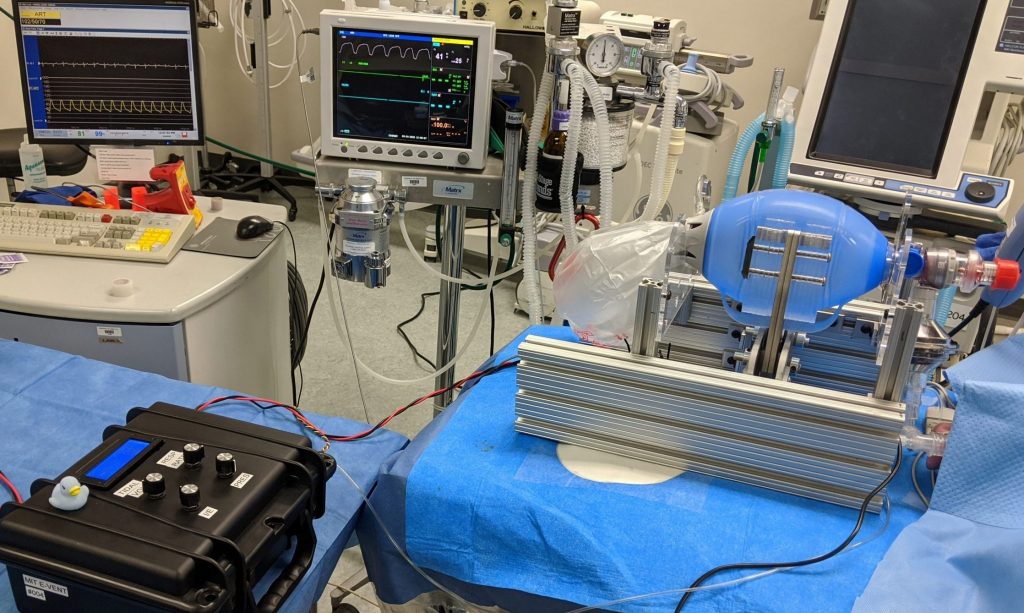
We have launched an emergency research project with a team of MIT Engineers and American clinicians to address the question:
Is it possible to safely ventilate a COVID-19 patient by automatically actuating a manual resuscitator?
Our process in approaching this question is to first identify the minimum requirements for a low-cost ventilator, based on the collective wisdom of many clinicians; design against these requirements; conduct immediate testing; report the results; iterate and facilitate discussion.
Manual ventilation is a short-term solution in a critical care environment, without any apparent clinical evidence regarding the safety of long-term use (days-weeks). There are multiple scenarios in which respiratory support could be needed: patients can be awake or asleep, sedated or sedated and paralyzed, breathing spontaneously, weaning off of a vent, etc. Furthermore, changing clinical presentations with ARDS require shifting minute ventilation (tidal volume ✕ respiratory rate) to “lung-protective” strategies, which place patients at risk for things like auto-PEEP. Some of these situations are simpler than others, with the simplest being ventilating a sedated, paralyzed patient. In such a situation, at a minimum a safe emergency ventilator could be used to free-up a conventional ventilator.
Any solution should be utilized only in a healthcare setting with direct monitoring by a clinical professional. While it cannot replace an FDA-approved ICU ventilator, in terms of functionality, flexibility, and clinical efficacy, the MIT Emergency Ventilator is anticipated to have utility in helping free up existing supply or in life-or-death situations when there is no other option.
Further, any low-cost ventilator system must take great care regarding providing clinicians with the ability to closely control and monitor tidal volume, inspiratory pressure, bpm, and I/E ratio, and be able to provide additional support in the form of PEEP, PIP monitoring, filtration, and adaptation to individual patient parameters. We recognize, and would like to highlight for anyone seeking to manufacture a low-cost emergency ventilator, that failing to properly consider these factors can result in serious long-term injury or death.
Open Source Design
At the present time, we are producing four sets of material, which we will be releasing and updating on this site in an open-source fashion:
- Minimum safe ventilator functionality based on clinical guidance
- Reference hardware design for meeting minimum clinical requirements
- Reference control strategies and electronics designs and supporting insights
- Results from testing in animal models
We are releasing this material with the intent to provide those with the ability to make or manufacture ventilators, the tools needed to do so in a manner that seeks to ensure patient safety. Clinicians viewing this site can provide input and expertise and report on their efforts to help their patients.
As with any research to design to scale-up to manufacture, we anticipated that there will be many problems and it is our goal to provide this site as a tool to “close the loop” and receive feedback. We will also do our best to publish the most relevant pieces of information in the discussion forum for all to see.
We invite anyone who is interested to follow this work.
Commenting is moderated and access to comment will be granted based on expertise and quality of comment. This is to ensure that the discussion is constructive and focused on safety.
Resources
For official information and recommendations on responding to COVID-19 please consult the following resources:
Information on MIT’s internal response and resources can be found here.
MIT, along with schools across the nation, has asked all of us to work from home. The crisis has created a unique opportunity to experiment in online / distance education and we are all learning together.
Thank you to the clinicians!
The MIT Emergency Ventilator Project team wishes to acknowledge the selfless contributions of the clinicians (and their supporting institutions), healthcare providers, and other professionals who have volunteered countless hours of their time to consult on this project, without compensation, as part of our community’s response to the COVID-19 emergency.
Support the fight against COVID-19
If you would like to help the people who are working hard to care for those affected by COVID-19, please consider donating to a reputable organization in your local community. Also we encourage all individuals to please think of your elderly or differently-abled neighbors who may need support. Other options to consider if you would like to help:
- Nationally, the American Red Cross is soliciting blood donations.
- The WHO has set up a response fund.
- The Department of Health and Human Services (DHHS) has declared liability immunity for medical countermeasures against COVID-19.
Help Support the MIT Team
If you would like to help support the all-volunteer MIT team, please consider making a donation. Donations to MIT are tax deductible. We are working long hours, spending fast and, because this project was launched in under a week, we are not a funded research project. You may donate directly below. For large donations, contact the team.

Terms of Use
We are endeavoring to provide our material in an open source format. Your use of this website and all Content contained herein is subject to the Terms of Use.
If you find this open-source material useful in your work, we will appreciate acknowledgment. Please note:
Use of MIT Name
Please note that “MIT”, “Massachusetts Institute of Technology”, and its logos and seal are trademarks of the Massachusetts Institute of Technology. Except for purposes of attribution as required pursuant to the Terms of Use, you may not use MIT’s names or logos, or any variations thereof, without prior written consent of MIT. You may not use the MIT name in any of its forms nor MIT seals or logos for promotional purposes, or in any way that deliberately or inadvertently claims, suggests, or in MIT’s sole judgment gives the appearance or impression of a relationship with or endorsement by MIT.
Latest updates
- COVID-19 Research SummaryWe have just launched a new page hosted by a team at the New York Medical College that lists and summarizes select, important peer-reviewed papers on the treatment of COVID-19.
- Waveform AnalysisIn this update we present a model-based and experimental analysis of the flow profiles of the MIT Emergency Ventilator for different patient conditions, incorporating ISO standards.
- Afghan Girls Robotics TeamWe excited that the Afghan Dreamers, an all-girls robotics team based in Afghanistan, has been inspired by the MIT Emergency Ventilator work and are building their own version. Congratulations to Roya Mahboob for bringing this team together. Read about it in Fast Company.
Recent Comments